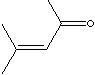
| MESITYL OXIDE | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PRODUCT IDENTIFICATION |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS NO. | 141-79-7 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| EINECS NO. | 205-502-5 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FORMULA | (CH3)2C=CHCOCH3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MOL WT. | 98.14 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H.S. CODE | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
TOXICITY |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SYNONYMS | Isobutenyl methyl ketone; Methyl isobutenyl ketone; | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1-Isobutenyl methyl ketone; 4-Methyl-3-penten-2-one; 4-Methylpent-3-en-2-one; Isopropylidene acetone; 4-Metilpent-3-en-2-ona; 4-Méthylpent-3-ène-2-one; 2-methyl-2-penten-4-one; | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
DERIVATION |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
CLASSIFICATION |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PHYSICAL AND CHEMICAL PROPERTIES |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PHYSICAL STATE | Clear liquid, with peppermint or honey-like odor | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MELTING POINT | - 59 C | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BOILING POINT |
130 C | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SPECIFIC GRAVITY | 0.865 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SOLUBILITY IN WATER | Slightly soluble | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| pH |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| VAPOR DENSITY | 3.39 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AUTOIGNITION |
344 C | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NFPA RATINGS | Health: 2; Flammability: 3; Instability: 1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
REFRACTIVE INDEX |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FLASH POINT |
30 C | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| STABILITY |
Stable at ordinary conditions. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
APPLICATIONS |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Methyl Isobutyl Ketone (MIBK) is a clear liquid with a mild characteristic odor;
miscible in oil, soluble in water. MIBK is a polar solvent. But the polarity is
similar to ethyl acetate. Water solubility is not good compare to other ketone
solvents like acetone and MEK (methyl ethyl ketone). This property makes MIBK an
useful liquid-liquid extraction solvent. MIBK is produced from acetone with
hydrogen by three-step process (aldol condensation, dehydration, hydrogenation).
Aiacetone alcohol (CAS #: 123-42-2) and mesityl oxide (CAS #: 141-79-7) are
intermediate products during the process. The basic unit quantity of acetone to produce 1
unit of MIBK is 1.22. MIBK has good compatibility with various organic reagents and
solvency power for a variety industrial materials. It is primarily used in
cellulose-based and resin-based coatings and adhesives. It is also employed in
rare-metal extraction. It is used in dewaxing to purify pharmaceuticals, mineral
oils, fatty acids, and alcohols. MIBK is also an useful intermediate to produce
target molecules, rubber antiozonants (e.g. 6PPD) and acetylenic diol compounds
are examples of end products.
Diacetone alcohol has slow evaporation rates. It is used as a solvent for both hydrogen bonding and polar substances. It is miscible in water and used as a solvent for water-based coatings. It is used as a solvent extractant in purification processes for resins and waxes. Diacetone alcohol is more suitable for use in applications as a component of gravure printing inks, with proving favorable flow and leveling characteristics. Diacetone alcohol, having hydroxyl and carbonyl group in the same molecule is used as a chemical intermediate. Mesityl oxide, a carbonyl compound having alpha (or beta) unsaturated chain, can be used as a raw material to produce drugs, solvents and plasticizer. Mesityl oxide is used to produce hydroperoxides. Mesityl oxide is as an extractant in ore flotation especially for actinide series elements (thorium and uranium). |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SALES SPECIFICATION | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| APPEARANCE |
Clear liquid | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
CONTENT (alpha) |
90.0% min | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ISOMER |
10.0% max | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WATER |
0.1% max | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ACIDITY |
0.02% max | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
COLOR, APHA |
40 max | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TRANSPORTATION | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PACKING | 180kgs in drum | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HAZARD CLASS | 3 (Packing Group: III) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| UN NO. | 1229 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OTHER INFORMATION | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| European Hazard Symbols: XN, Risk Phrases: 10-20/21/22, Safety Phrases: 9-16-25-33 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GENERAL DESCRIPTION OF SOLVENT | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Solvent is a substance, usually a liquid, that acts as a dissolving agent or
that is capable of dissolving another substance. In solutions of solids or gases
in a liquid, the liquid is the solvent. In all other homogeneous mixtures (i.e.,
liquids, solids, or gases dissolved in liquids; solids in solids; and gases in
gases), solvent is the component of the greatest amount. The minor proportion
substances are called solutes. The solvent offers several functions during a
chemical reaction. It solves not only the substance that reacts with another one
to produce a new set of substances (reactant) but also the compound that
supplies the molecule, ion, or free radical, which is considered as the
attacking species in a chemical reaction (reagent). The solvent is conductive to
collisions between the reactants and reagents to transform the reactants to new
products. The solvent also takes roll of temperature control, either to provide
the energy of the colliding particles for speedy reaction and to absorb heat in
exothermic reaction. The appropriate solvent should be selected based on the
inactivity in the reaction conditions, dissolving the reagents as well as
reactants, appropriate boiling point and easy removal at the end of the
reaction.
Polarity The most common solvent is water. Other common solvents which dissolve substances that are insoluble (or nearly insoluble) in water are acetone, alcohol, formic acid, acetic acid, formamide. BTX, carbon disulfide, diemthyl sulfoxide, carbon tetrachloride, chloroform, ether, tetrahydrofuran, furfural, hexane and turpentine. They may be classified as polar and non-polar. Polar solvents, like water, have molecules whose electric charges are unequally distributed, leaving one end of each molecule more positive than the other. Usually polar solvent has O-H bond of which water (HOH), (CH3OH) and acetic acid (CH3COOH) are examples. Propanol, butanol, formic acid, formamide are polar solvents. Dipolar solvents which contain a C-O double bond without O-H bond are acetone [(CH3)2C=O], ethyl acetate (CH3COOCH2CH3), methyl ethyl ketone, acetonitrile, N,N-dimethylformamide and diemthyl sulfoxide. Nonpolar solvents, like carbon tetrachloride (CCl4), benzene (C6H6), and diethyl ether ( CH3CH2OCH2CH3), have molecules whose electric charges are equally distributed and are not miscible with water. Hexane, tetrahydrofuran and methylene chloride are non-polar solvents. Polar solvents are hydrophilic but non-polar solvents are lipophilic. Polar reactants will dissolve in polar solvents. Non-polar solvents dissolve non-polar compounds best. Oil and water don't mix but separate into two layers. There are three measures of the polarity as "dipole moment", "dielectric constant" and "miscibility with water". Though low dipole moments and small dielectric constants indicates non-polar solvents, sharp boundaries between polar and non-polar solvents are not available. The polarity reflects the balance between a polar component (OH) and a non-polar hydrocarbon component, existing in the same molecule. If hydrocarbon character increases relatively, the polarity decreases. On an operational basis, solvents that are miscible with water are polar. Polar Protic and Dipolar Aprotic Protic refers to a hydrogen atom attached to an electronegative atom. Protic solvents can donate an H+ (proton) since they contain dissociable H+, such as hydrogen attached to oxygen as in a hydroxyl group, nitrogen as in a amine group. Examples are water, methanol, ethanol, formic acid, hydrogen fluoride and ammonia. Aprotic solvents don't has O-H bond but a C=O bond typically. Examples are acetone [(CH3)2C=O] and ethyl acetate (CH3COOCH2CH3). Polar protic solvents are useful in SN1 reaction, while polar aprotic solvents are SN2 reaction.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||